DID YOU KNOW... Pheomelanin and Eumelanin have completely different pathways of formation.
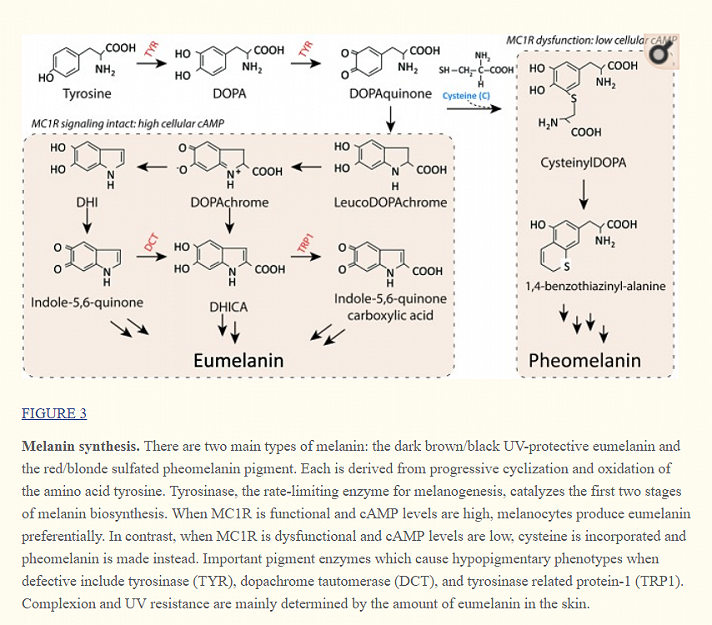
"Pheomelanin, consists mainly of sulfur-containing benzothiazine and benzothiazole derivatives.
L-cysteine is the chief source of sulfur and is, therefore, essential for the pheomelanin synthesis.
Eumelanin, on the other hand is an extremely heterogeneous polymer comprising of 5,6-dihydroxyindole (DHI) and or 5,6-dihydroxyindole-2-carboxylic acid (DHICA) units.
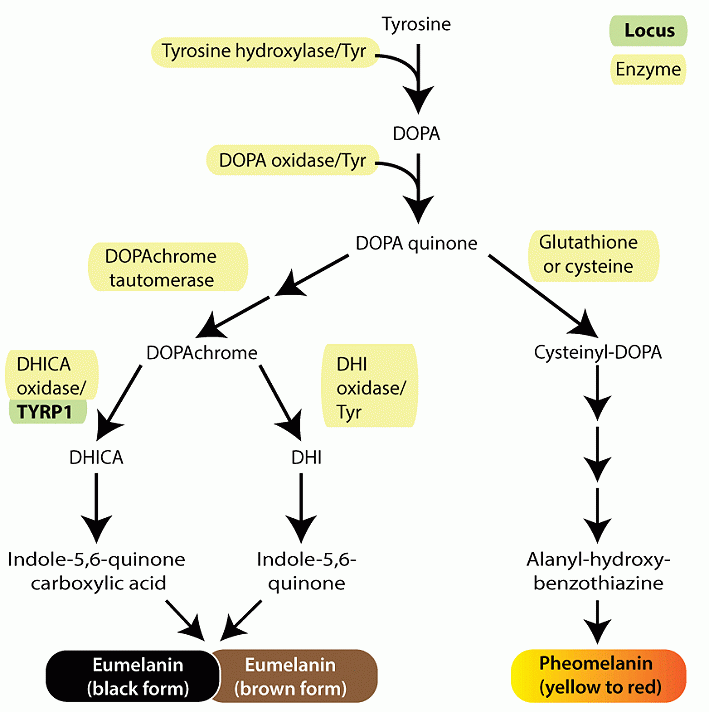
Both eumelanin and pheomelanin derive from the sequential cyclization and oxidation of the amino acid tyrosine.
The first two biosynthetic steps are shared between the two pathways: the conversion of tyrosine to DOPA and then to DOPAquinone by the enzyme tyrosinase.
Eumelanogenesis and pheomelanogenesis diverge after formation of DOPAquinone.
Other enzymes beside tyrosinase are needed for melanin synthesis including dopachrome tautomerase and tyrosinase-related protein 1.
Defects in many pigment enzymes yield hypomelanotic phenotypes such as albinism.
Pheomelanin production is dependent upon the incorporation of a cysteine and retention of sulfur after the synthesis of DOPAquinone, which may explain why mature pheomelanin pigments are reddish/yellow rather than dark brown/black as eumelanin is.
Although the control of the pigment switch between eumelanin and pheomelanin is regulated by multiple factors including the pH of the cellular milieu and the levels of tyrosinase (Burchill and Thody, 1986; Ancans et al., 2001), the presence of a functional MC1R is required for effective synthesis of eumelanin.
Since eumelanin absorbs UV radiation, the more eumelanin the skin has, the more protected it is from UV damage."
"Pheomelanin, consists mainly of sulfur-containing benzothiazine and benzothiazole derivatives.
L-cysteine is the chief source of sulfur and is, therefore, essential for the pheomelanin synthesis.
Eumelanin, on the other hand is an extremely heterogeneous polymer comprising of 5,6-dihydroxyindole (DHI) and or 5,6-dihydroxyindole-2-carboxylic acid (DHICA) units.
Both eumelanin and pheomelanin derive from the sequential cyclization and oxidation of the amino acid tyrosine.
The first two biosynthetic steps are shared between the two pathways: the conversion of tyrosine to DOPA and then to DOPAquinone by the enzyme tyrosinase.
Eumelanogenesis and pheomelanogenesis diverge after formation of DOPAquinone.
Other enzymes beside tyrosinase are needed for melanin synthesis including dopachrome tautomerase and tyrosinase-related protein 1.
Defects in many pigment enzymes yield hypomelanotic phenotypes such as albinism.
Pheomelanin production is dependent upon the incorporation of a cysteine and retention of sulfur after the synthesis of DOPAquinone, which may explain why mature pheomelanin pigments are reddish/yellow rather than dark brown/black as eumelanin is.
Although the control of the pigment switch between eumelanin and pheomelanin is regulated by multiple factors including the pH of the cellular milieu and the levels of tyrosinase (Burchill and Thody, 1986; Ancans et al., 2001), the presence of a functional MC1R is required for effective synthesis of eumelanin.
Since eumelanin absorbs UV radiation, the more eumelanin the skin has, the more protected it is from UV damage."